What is Water Conductivity? A Complete Guide to Conductivity Meters
📊 Water Quality Hub 📅 March 14, 2025 ⏱ 9 min read ⚡ conductivity · TDS · sensors
💧 The hidden conductor in water — Pure distilled water is almost an electrical insulator. So what turns ordinary water into a conductor? The answer lies in dissolved ions. This article demystifies water conductivity, explores why it matters, and delivers a deep dive into the working principle of conductivity meters — from sensor design to signal processing and temperature compensation.
1. What is Water Conductivity?
Electrical conductivity (EC) measures the ability of a water sample to carry an electrical current. It is expressed in microsiemens per centimeter (µS/cm) or millisiemens per centimeter (mS/cm). The more ions dissolved in water (e.g., sodium, calcium, chloride, sulfate), the higher the conductivity.
1.1 Conductivity vs. resistivity
Conductivity is the reciprocal of resistivity. Ultra-pure water has resistivity around 18.2 MΩ·cm (conductivity ≈ 0.055 µS/cm). Tap water ranges from 100–800 µS/cm, while seawater can exceed 50 mS/cm due to high Na⁺ and Cl⁻ concentrations.
1.2 Key factors affecting conductivity
-
Ion concentration & charge: More ions and higher charge (Ca²⁺, Mg²⁺) increase conductivity.
-
Temperature: Conductivity rises about 1.9% per °C (depending on ion type). Modern meters include automatic temperature compensation (ATC) to normalize readings to 25°C.
-
Pollutants: Industrial or agricultural runoff introduces extra ions, altering conductivity dramatically.
2. Why Measure Water Conductivity?
Conductivity is a rapid, cost‑effective indicator for:
-
Purity monitoring: Pharma, semiconductor rinsing, boiler feedwater.
-
Environmental assessment: Sudden changes in rivers/lakes may indicate pollution or salt intrusion.
-
Agriculture & aquaculture: Irrigation water quality, fish tank total dissolved solids (TDS) correlate with EC.
-
Industrial processes: Cooling towers, wastewater treatment — high conductivity can cause scaling or corrosion.
3. Conductivity Meter Working Principle — Full Breakdown
A conductivity meter consists of a sensor (electrode) and a transmitter / main unit that processes the signal. The principle is based on Ohm’s law: measure the resistance between two electrodes immersed in the sample, then convert to conductivity.
But raw conductance depends on electrode geometry — that’s where the cell constant (K) comes in.
3.1 The cell constant (K) — the missing factor
Conductivity (γ) = measured conductance (G) × cell constant (K). K = distance between electrodes (L) / effective area (A), unit cm⁻¹. Different K values suit different ranges: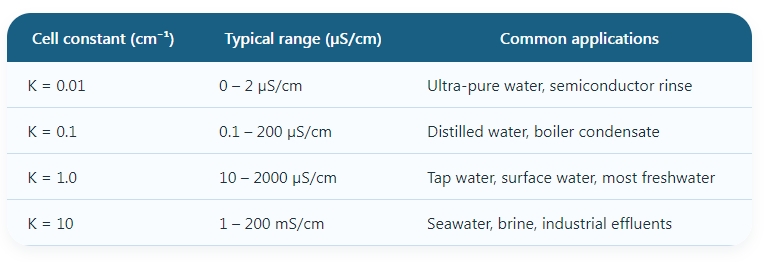
Most meters allow calibration with standard KCl solutions and automatic cell constant recognition.
3.2 From analog signal to display: the signal chain
The tiny electrode current is converted to voltage (I/V converter), then rectified, filtered, and often phase‑sensitive detected to eliminate capacitive interference. After ADC conversion, a microprocessor applies temperature compensation and displays the value normalized to 25°C.
3.3 Advanced: four‑electrode and inductive sensors
-
Four‑electrode technology: Two electrodes carry current, two separate electrodes measure voltage drop. This eliminates errors from cable resistance, electrode fouling, and polarization — ideal for high‑conductivity or dirty samples.
-
Inductive (toroidal) sensors: Two encapsulated coils. One induces a current in the sample loop, the other senses the induced signal. No metal contacts with liquid, perfect for aggressive chemicals or slurries.
4. Choosing the Right Conductivity Meter
Based on your application:
-
Benchtop meters: High precision, multiple ranges, often with stirring function — for labs and QA.
-
Portable / handheld: Field surveys, spot checks; rugged and waterproof.
-
Inline / industrial sensors: Mounted in pipes or tanks with 4‑20 mA output, often with high temperature/pressure ratings.
🧪 Pro tip: Always calibrate with fresh standard solution; keep electrodes clean and hydrated. For ultra‑pure water, use a flow‑cell to prevent CO₂ absorption from air, which raises conductivity.
Conclusion
Water conductivity is a fast, reliable proxy for total ion content — essential for industries from pharmaceuticals to environmental science. Behind every accurate reading is a conductivity meter that cleverly applies AC excitation, advanced electrode design, and precise signal processing to turn a simple electrical measurement into a meaningful water quality parameter. Understanding its working principle helps you choose the right instrument, interpret data correctly, and maintain your sensor for years of reliable service.