1. Introduction: What is pH?
pH (potential of hydrogen) is a measure of hydrogen ion concentration in a solution, indicating its acidity or alkalinity. The pH scale ranges from 0 to 14, with 7 being neutral. Values below 7 indicate acidity, while values above 7 indicate alkalinity. Each unit change represents a tenfold change in hydrogen ion concentration.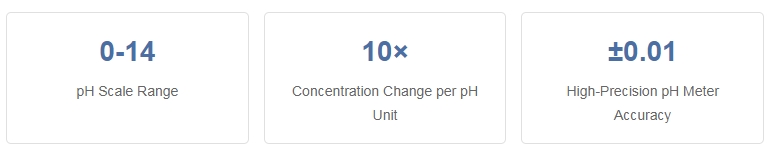
Where [H⁺] is the hydrogen ion concentration in moles per liter
The pH meter is an electrochemical device that measures the activity of hydrogen ions in a solution. Unlike simple indicators that change color, a pH meter provides precise, quantitative measurements essential for scientific research, industrial quality control, and environmental monitoring.
2. The Glass Electrode: Heart of the pH Meter
The glass electrode is the core sensing element of a pH meter. It consists of a special glass membrane that is selectively sensitive to hydrogen ions. When immersed in a solution, the glass membrane develops an electrical potential proportional to the hydrogen ion activity difference between the inside and outside of the membrane.
┌─────────────────────────────────────┐
│ Reference Electrode (Ag/AgCl) │
│ ┌─────────────────────────────┐ │
│ │ Internal Reference Solution │ │
│ │ (pH 7 buffer) │ │
│ ├─────────────────────────────┤ │
│ │ Glass Membrane │ │
│ │ (pH-sensitive) │ │
│ └─────────────────────────────┘ │
│ ↓ │
│ Test Solution │
└─────────────────────────────────────┘
Composition: Special lithium or sodium silicate glass with high hydrogen ion selectivity
Thickness: 0.03-0.1 mm
Function: Develops potential proportional to H⁺ activity difference
Type: Silver/Silver chloride (Ag/AgCl) or Calomel (Hg/Hg₂Cl₂)
Function: Provides stable internal potential
Electrolyte: 3M KCl solution with pH 7 buffer
Composition: pH 7 buffer with fixed chloride concentration
Function: Maintains constant internal pH and chloride activity
Replenishment: Refillable or sealed gel types available
2.1 How the Glass Electrode Works
The glass membrane develops a potential (Eₘ) that follows the Nernst equation:
Where:
E₀ = standard potential
R = gas constant (8.314 J/mol·K)
T = absolute temperature (K)
n = number of electrons transferred (1 for H⁺)
F = Faraday constant (96,485 C/mol)
a_H⁺ = hydrogen ion activity
When the glass membrane separates two solutions with different hydrogen ion activities, a potential difference develops. This potential is linearly related to the logarithm of the hydrogen ion activity ratio.
3. Reference Electrode: The Stable Reference Point
The reference electrode provides a stable, constant potential against which the glass electrode potential is measured. Without a stable reference, accurate pH measurement would be impossible.
3.1 Types of Reference Electrodes
| Type | Reference System | Advantages | Disadvantages |
|---|---|---|---|
| Ag/AgCl | Silver wire coated with AgCl in KCl | Stable, wide temperature range, non-toxic | Silver ions can contaminate some samples |
| Calomel (Hg/Hg₂Cl₂) | Mercury in contact with calomel in KCl | Very stable, historically common | Mercury toxicity, temperature sensitive |
| Double Junction | Two electrolyte chambers to prevent contamination | Prevents reference electrolyte from entering sample | Higher cost, slower response |
3.2 Liquid Junction and Salt Bridge
The liquid junction is the point where the reference electrode electrolyte contacts the sample solution. A porous ceramic frit or fiber allows ionic contact while preventing rapid mixing. Common junction types include:
- Ceramic frit: Durable, moderate flow rate
- PTFE sleeve: High flow rate, self-cleaning
- Open junction: Best for low ionic strength samples
- Ground glass sleeve: Adjustable flow, easy cleaning
4. The Complete pH Measurement System
A complete pH measurement system consists of three main components working together:
Critical Design Feature: The pH meter must have extremely high input impedance (10¹² Ω or higher) to measure the glass electrode potential without loading it. Standard voltmeters cannot measure this signal directly due to the electrode's very high internal resistance.
5. The Nernst Equation and Temperature Effects
The relationship between measured potential and pH is described by the Nernst equation. At 25°C (298 K), the equation simplifies to:
Where 0.05916 V is the Nernst slope at 25°C
5.1 Temperature Dependence
The Nernst slope changes with temperature according to:
| Temperature (°C) | Nernst Slope (mV/pH) | Temperature Effect on pH |
|---|---|---|
| 0 | 54.20 | ±0.03 pH per °C |
| 20 | 58.16 | ±0.03 pH per °C |
| 25 | 59.16 | Reference point |
| 30 | 60.15 | ±0.03 pH per °C |
| 40 | 62.13 | ±0.03 pH per °C |
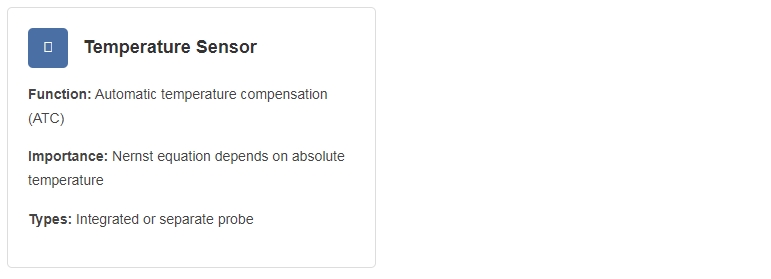
Automatic Temperature Compensation (ATC) is essential for accurate pH measurement. The meter continuously adjusts the slope calculation based on the measured temperature of the sample.
6. Calibration: The Key to Accurate pH Measurement
Calibration establishes the relationship between the measured electrode potential and actual pH. Modern pH meters use multi-point calibration with standard buffer solutions to determine both the electrode's slope and offset.
6.1 Calibration Procedure
- Rinse the electrode with distilled water
- Immerse in pH 7.00 buffer and adjust the offset (asymmetry potential)
- Rinse and immerse in pH 4.01 or pH 10.01 buffer
- Adjust the slope (sensitivity)
- Verify with a third buffer point (optional)
6.2 Calibration Standards
| Buffer Type | pH at 25°C | Common Use |
|---|---|---|
| Oxalate | 1.68 | Acidic range calibration |
| Phthalate | 4.01 | Primary acidic buffer |
| Phosphate | 6.86 / 7.00 | Neutral range calibration |
| Borate | 9.18 / 10.01 | Alkaline range calibration |
| Carbonate | 12.45 | Highly alkaline calibration |
Calibration is required before each use or at regular intervals. Electrodes can drift over time due to membrane aging, contamination, or reference junction issues. A properly functioning electrode should achieve a slope between 95% and 105% of theoretical (about 56-62 mV/pH at 25°C).
7. Electrode Types and Special Applications
7.1 Electrode Configurations
- Combination Electrodes: Most common design, combining glass and reference in one body
- Separate Electrodes: Used in specialized applications where flexibility is needed
- Microelectrodes: For small sample volumes (as low as 50 μL)
- Flat Surface Electrodes: For semi-solid materials (food, skin, paper)
- Spear Tip Electrodes: For piercing solids like cheese, meat, or soil
7.2 Specialized Electrodes
| Type | Application | Key Feature |
|---|---|---|
| High-Temperature | Industrial processes, sterilization | Operates up to 130°C |
| Low-Ionic Strength | Deionized water, rainwater | Special junction design for high resistance |
| High-Alkaline | Wastewater, cleaning solutions | Resistant to sodium error (pH > 12) |
| High-Fouling | Food processing, bioreactors | Self-cleaning or easy-clean designs |
| Antimony | High-temperature, harsh environments | Metal electrode, no glass breakage |
8. Common Measurement Errors and Troubleshooting
8.1 Common Problems and Solutions
| Problem | Possible Causes | Solutions |
|---|---|---|
| Slow response / Drifting | Contaminated electrode, aged membrane, dirty junction | Clean electrode, rehydrate, replace if necessary |
| Erratic readings | Loose connection, electrostatic interference, damaged cable | Check connections, use shielded cable, ensure proper grounding |
| Slope outside 95-105% | Aged electrode, contaminated reference, expired buffers | Clean thoroughly, recalibrate with fresh buffers, replace electrode |
| Offset > ±30 mV | Electrode aging, reference junction problems | Clean junction, restore reference electrolyte, replace electrode |
| Cannot calibrate | Dead electrode, faulty meter, contaminated buffers | Check with known good electrode, replace buffers |
8.2 Sodium Error (Alkaline Error)
At high pH values (>12), glass electrodes can respond to sodium ions instead of hydrogen ions, causing measurement errors. Special low-sodium-error glass formulations minimize this effect.
8.3 Acid Error
At very low pH (<1), the glass membrane can exhibit non-linear response, requiring specialized electrodes for accurate measurement.
9. Electrode Maintenance and Storage
9.1 Proper Storage
- Short-term: Store in pH 4 or 7 buffer solution (never in distilled water)
- Long-term: Store with protective cap containing storage solution or buffer
- Never: Store dry or in distilled water (removes hydration layer)
9.2 Cleaning Procedures
- General cleaning: Rinse with distilled water between measurements
- Protein contamination: Soak in pepsin/HCl solution
- Oil/grease: Wash with mild detergent solution
- Inorganic deposits: Soak in 0.1 M HCl
- Bacterial growth: Soak in dilute bleach solution
9.3 Electrode Rehydration
If a glass electrode dries out, the hydration layer is lost. Rehydrate by soaking in pH 4 buffer for 24-48 hours before use.
Electrode Lifespan:
- General purpose electrodes: 1-2 years with proper care
- Heavy use / harsh conditions: 6-12 months
- Signs of aging: slow response, drift, low slope, inability to calibrate
10. Modern Advances in pH Measurement Technology
10.1 ISFET pH Sensors
Ion-Sensitive Field Effect Transistors (ISFET) offer an alternative to glass electrodes. These solid-state sensors are:
- Breakage-resistant (no glass)
- Faster response time
- Compatible with gel-filled reference systems
- Require no hydration (always ready to use)
10.2 Optical pH Sensors
Fluorescence-based pH sensors use indicator dyes immobilized in a matrix. Applications include:
- Non-invasive measurement (through container walls)
- Microfluidic systems
- Bioreactor monitoring
- In vivo medical applications
10.3 Wireless and Smart pH Meters
Modern pH meters feature:
- Bluetooth connectivity for data logging and mobile apps
- Cloud-based data storage and analysis
- Automatic calibration reminders
- Self-diagnostic capabilities
Future developments in pH sensing focus on miniaturization, wireless connectivity, and integration with industrial IoT (IIoT) systems for real-time process monitoring and control.